ENRICH YOUR MIND
AND BODY
Enrich Your
Mind And Body
Upgrade your wellness at our integrative
center for functional medicine.
Upgrade your wellness at our integrative center for functional medicine.
Our Integrative Approach to health

Take control of your well-being now.
Start Reaching Optimal Health Today.
Recharge your cellular vitality. Improve your quality of life.
Promote healthy longevity by heightening your metabolism,
cognition, hormones, immunity and anti-inflammatory
functions. Uncover the underlying connections between
your different body systems.
Recharge your cellular vitality. Improve your quality of life. Promote healthy longevity by heightening your metabolism, cognition, hormones, immunity and anti-inflammatory functions. Uncover the underlying connections between your different body systems.
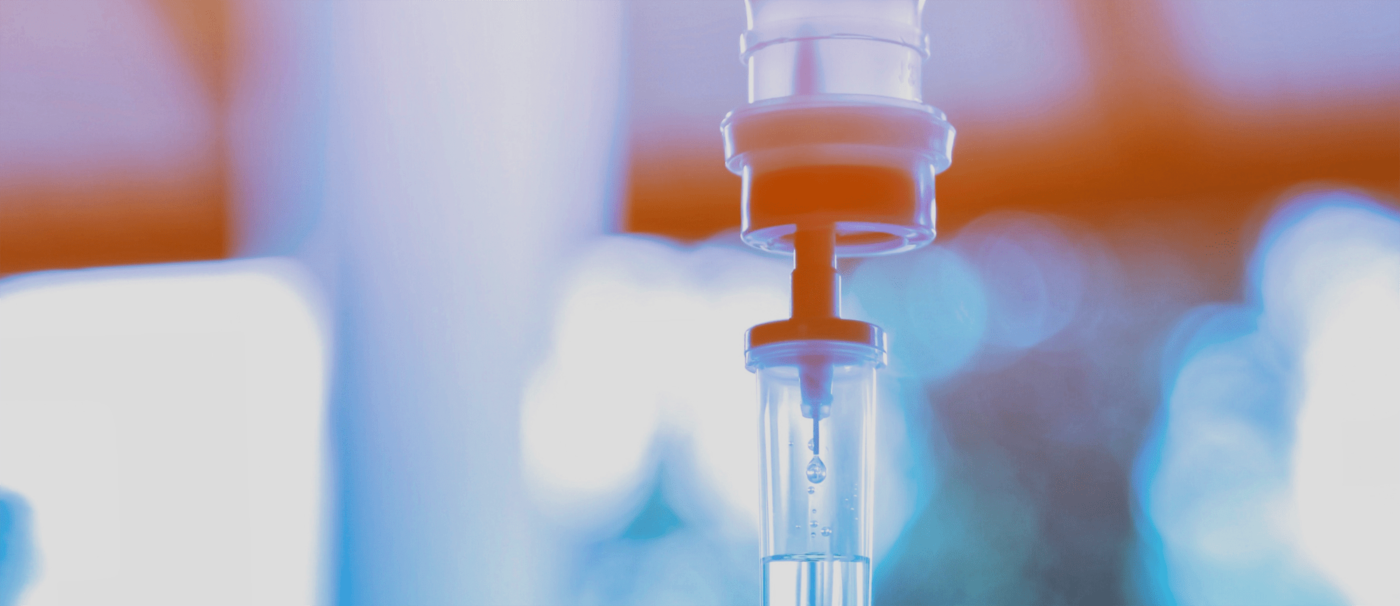
The IV Lounge
Upgrade yourself with restorative IV infusions. Our IV drips work synergistically with tailored functional medicine therapies to optimize your energy levels. IV drips may offer brain care support, while helping to correct digestive shortcomings, prevent illness, and promote healthy longevity.
THE IV LOUNGE
Upgrade yourself with restorative IV infusions. Our IV drips work synergistically with tailored functional medicine therapies to optimize your energy levels. IV drips may offer brain care support, while helping to correct digestive shortcomings, prevent illness, and promote healthy longevity.
OUR LONGEVITY
APPROACH TO WELLNESS
Our longevity approach to wellness has three pillars – gut health, brain health and hormonal balance. Together these three pillars address root cause, inflammation, optimize immune function and cellular health to achieve optimal health and well-being.
OUR LONGEVITY
APPROACH TO WELLNESS
Our longevity approach to wellness has three pillars – gut health, brain health and hormonal balance. Together these three pillars address root cause, inflammation, optimize immune function and cellular health to achieve optimal health and well-being.

OUR LONGEVITY
APPROACH TO WELLNESS
Our longevity approach to wellness has three pillars – gut health,
brain health and hormonal balance. Together these three pillars address
root cause, inflammation, optimize immune function and cellular
health to achieve optimal health and well-being.

BLOG / THE IV LOUNGE
We provide education to patients and readers on the integrative functional medicine approach to healthcare, showing how it can contribute to your journey toward optimal wellness.
Explore our blog here.
We provide education to patients and readers on the integrative functional medicine approach to healthcare, showing how it can contribute to your journey toward optimal wellness. Explore our blog here.
Explore Functional Medicine: Your Path to Optimal Health and Well-Being
BLOG / THE IV LOUNGE
EXPLORE FUNCTIONAL MEDICINE: YOUR PATH TO OPTIMAL HEALTH AND WELL-BEING
We provide education to patients and readers on the integrative functional medicine approach to healthcare, showing how it can contribute to your journey toward optimal wellness. Explore our blog here.





